10.3 Chemical Kinetics
10.3.1 Reaction Rate
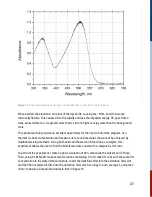
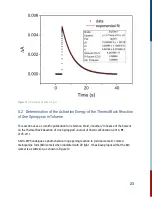
The rate of a chemical reaction is expressed as the variation with time of the concentration of
either reactants or products. In solution phase reactions, as studied here, the units of reaction rate
are concentration units per second, i.e. mole per liter per second, M s .
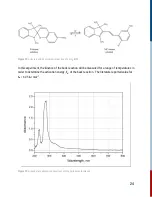
In the (elementary) reaction,
A + B
⟶
C
(2)
The rate (change in concentration as a function of time) is expressed in the form of a derivative
(3)
10.3.2 Rate Expression and Rate Constant
The rate expression containing a constant of proportionality or rate constant, k, is
(8)
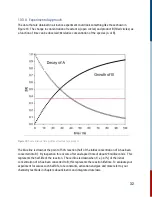
This equation tells us that since the concentrations of the reactants decrease during the reaction,
then the rate (but not the rate constant) decreases with time elapsed. Thus, the rate constant
provides the spectroscopist with a useful measure of the velocity of a reaction, an important
quantity to determine when studying chemical kinetics.
10.3.3 Order of Reaction
In the previous rate equation (Equation 8) the concentrations of A and B both appear to the first
power, i.e., this reaction is first order with respect to both [A] and [B], and overall second order
reaction.
In the dimerization reaction for the elementary process,
2A
⟶
C
(4)
the rate expression is will be
(9)
−
1
Rate
=
−
d
[
A
]
dt
=
−
d
[
B
]
dt
=
d
[
C
]
dt
−
d
[
A
]
dt
=
k
[
A
][
B
]
−
d
[
A
]
dt
=
k
[
A
][
A
] =
k
[
A
]
2
30