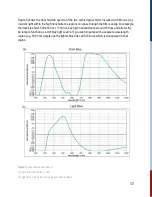
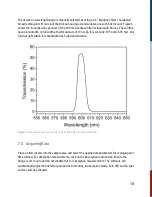
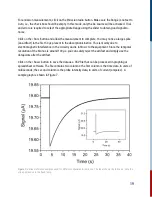
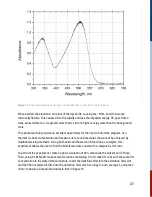
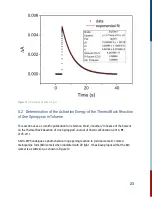
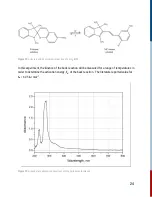
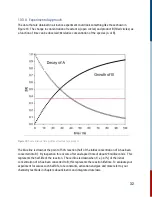
For this reaction, it is second order in [A] and overall second order.
Also, the symbolic elementary process
A
⟶
P
(10)
would be expected to have a rate equation
(11)
and the reaction is first order in [A] and overall first order. Orders higher than 2, fractional and zero
orders are found in special cases. These are not considered here because they are not relevant to
Kronos.
NOTE:
It is important to realize that assignment of order by inspection is only valid for elementary
reactions; it does not apply for stoichiometric equations.
−
d
[
A
]
dt
=
k
[
A
]
31